IonDX is the only life sciences instrument company manufacturing bench-top atmospheric ion mobility spectrometers for native biomolecules
Measurement Principle
Gas-phase ion collision cross-sectional area via native electrospray ion mobility
IonDX measures SINGLY charged analyte ions avoiding the well-established correlation between charge state and protein unfolding in the gas phase, while making DIRECT measurements of collisional cross-sections.
Accelerate Biotherapeutics Development
- Protein engineering
- Candidate selection
- Biological stability
- Sample screening
- Biosimilarity
- Bioprocess optimization
- Batch/batch comparability
Modality Agnostic
- Monoclonal antibodies
- Non-covalent protein complexes
- Glycoproteins
- CRISPR-Cas
- >1MDa mRNA and mRNA-LNPs
- Plasmid DNAs
- AAVs
Expand Your Totality of Evidence
Drug concentration
Binding
Purity
Higher order structure
Conformational stability
Protein screening
- 100% Electrospray Ion Mobility Focused
Unlock an orthogonal method for HOS characterization

Rapid Scans of mAb Higher Order Structure
mAb HOS data in less than 2 minutes
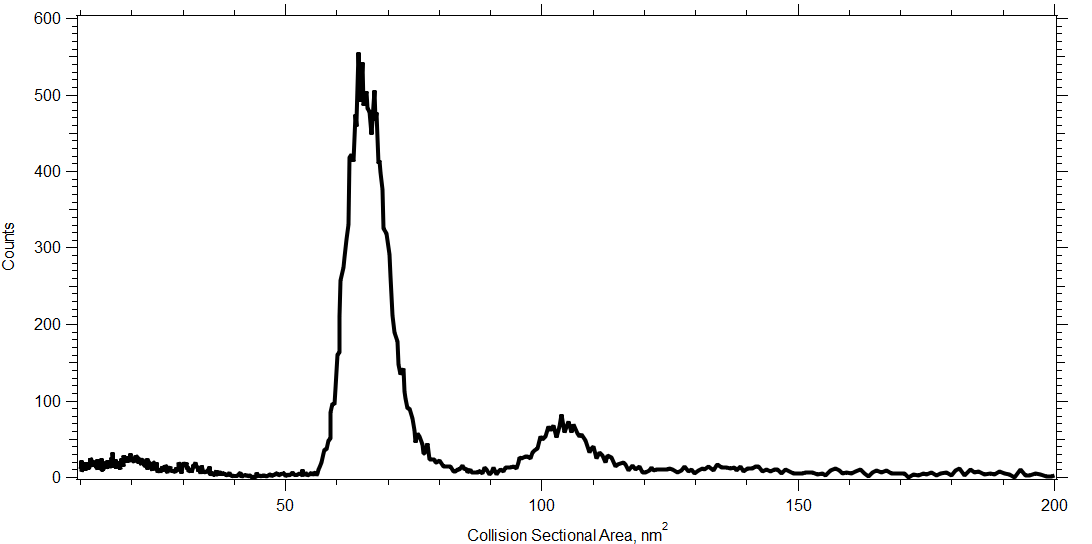
mAbs have fluctuating & dynamic higher order structure in solution, including the presence of oligomers. We measure the cross-sectional area of all those conformations in <2 min with femtomole sensitivity. Typical samples are 0.05 mg/ml and we consume 100 nl during a single scan. In this single scan plot you see the baseline resolution of the NISTmAb monomer and dimer.

Investigate mAb Oligomers
Quantitative direct measurement of macromolecules
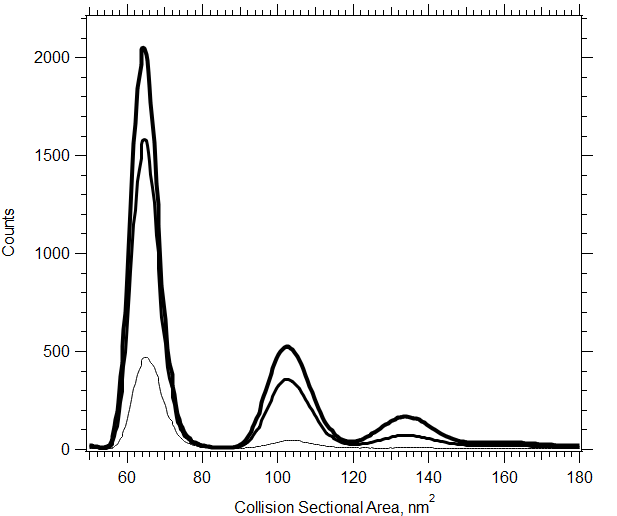
Ion mobility provides you with a rapid means of surveying not only intact mAbs, but their non-specific aggregates and other higher order structures formed in solution. Common applications include ADC characterization and forced degradation pathways. This plot reveals conformations of singly-charged NISTmAb monomers, dimers and trimers with increased concentration. The m/z of these singly-charged ions extends beyond 450,000 precluding detection by standard mass spectrometers.
Fenn introduced proteins into the gas phase.
Then we argued about their conformation for 30 yrs.
Finally, measurements not distorted by electrospray.
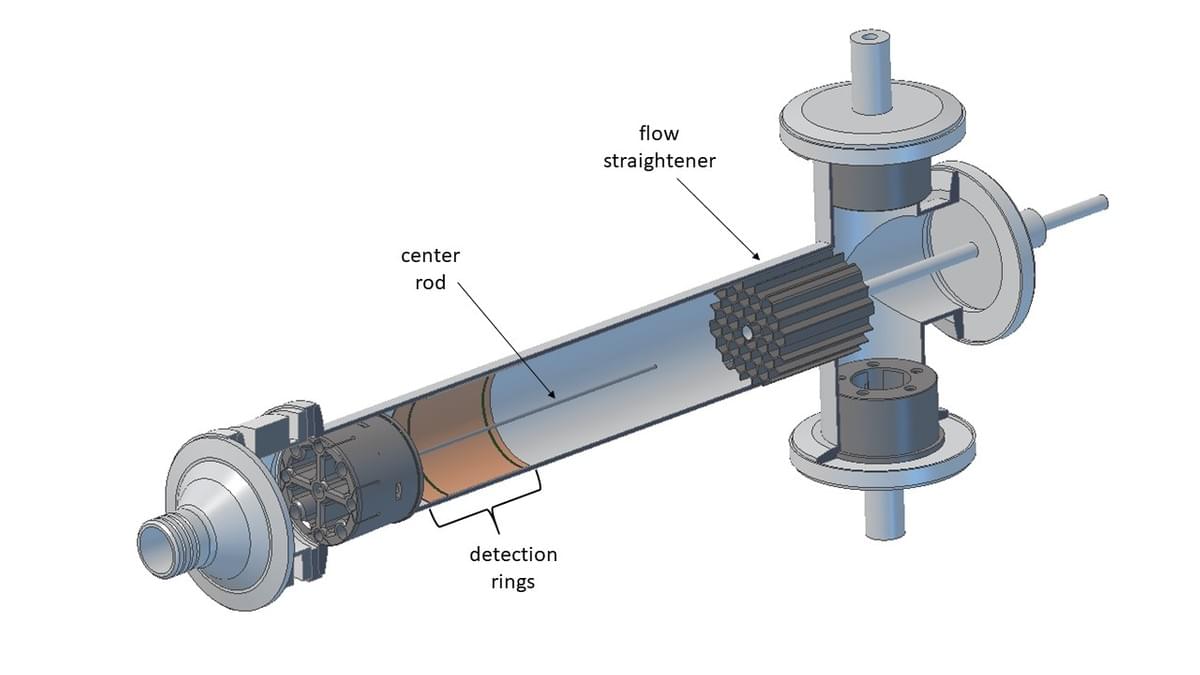
We measure gas phase cross-sections of intact singly-charged macromolecules
using our patented ion mobility spectrometer
IT'S SIMPLE.

Our measurements cannot be made with any other instrument.
We measure the size & shape (conformation) of giant biomolecules via a DIRECT ion mobility method that uses a combination of aerodynamics and electric fields.
Curious?
You know you want to learn more.
Native Electrospray Ion Mobility of your Macromolecules is at your fingertips!
About Us
Our Mission
We're Hiring!
Resources
Tutorials
Brand Assets
Contact Us
info@